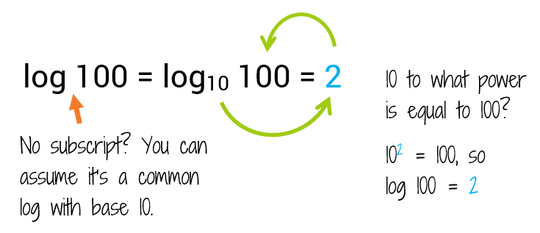Find the logarithm of : (i) 100 to the base 10 (ii) 0.1 to the base 10 (iii) 0.001 to the base 1... - YouTube
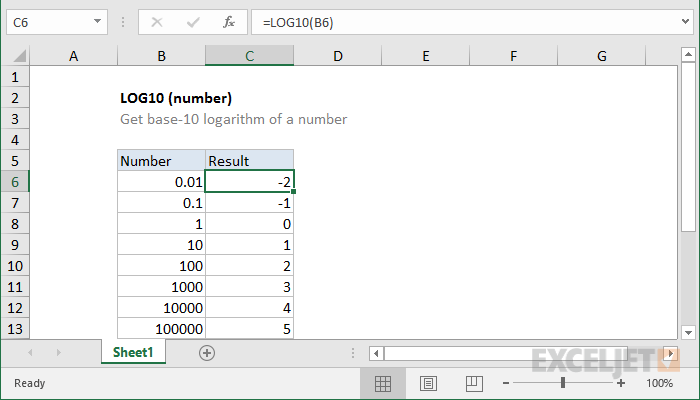
![SOLVED: Acidic or basic solution, pH The pH of a solution is the negative logarithm of the hydrogen-jon concentration: pH = #log[h"] log stands for logarithm to the base 10 log 100 = SOLVED: Acidic or basic solution, pH The pH of a solution is the negative logarithm of the hydrogen-jon concentration: pH = #log[h"] log stands for logarithm to the base 10 log 100 =](https://cdn.numerade.com/ask_images/54471c304890403dbf6cbad11744b807.jpg)
SOLVED: Acidic or basic solution, pH The pH of a solution is the negative logarithm of the hydrogen-jon concentration: pH = #log[h"] log stands for logarithm to the base 10 log 100 =