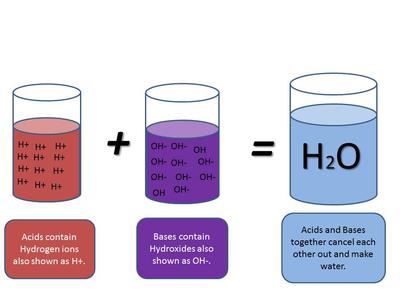
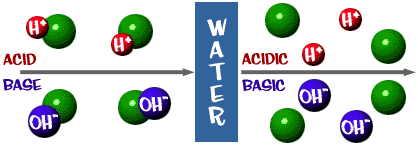
SOLVED: Acids ionize in water to produce H+. This H+ causes indicators like phenolphthaleinand litmus to change color. The un-ionized acid molecules have no effect on indicators.Ionic equations – total and net -

Is (half equivalence point) when a conjugate base and an acid are equal in concentration. But (equivalence point) is when H+ = OH-? And is ph=pka at half equivalence point but not






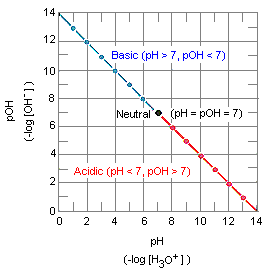
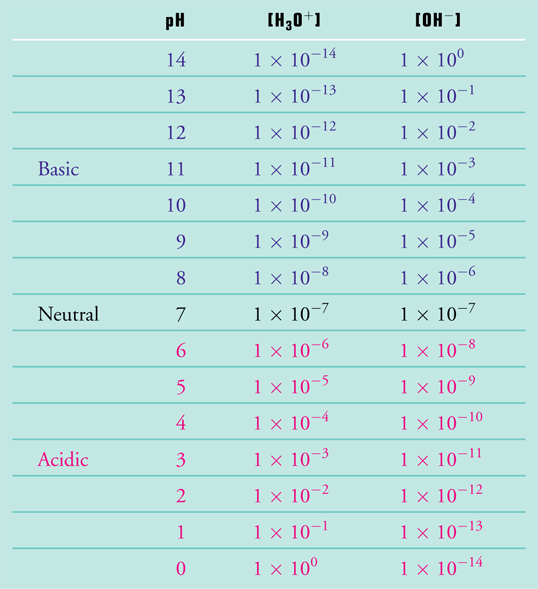
![Calculating pH, pOH, [H+], [OH-] - Acids and Bases Calculating pH, pOH, [H+], [OH-] - Acids and Bases](http://iloveacid--basechemistry.weebly.com/uploads/2/7/8/0/27808151/2222607_orig.gif)

![Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube](https://i.ytimg.com/vi/UiK37I159fc/maxresdefault.jpg)
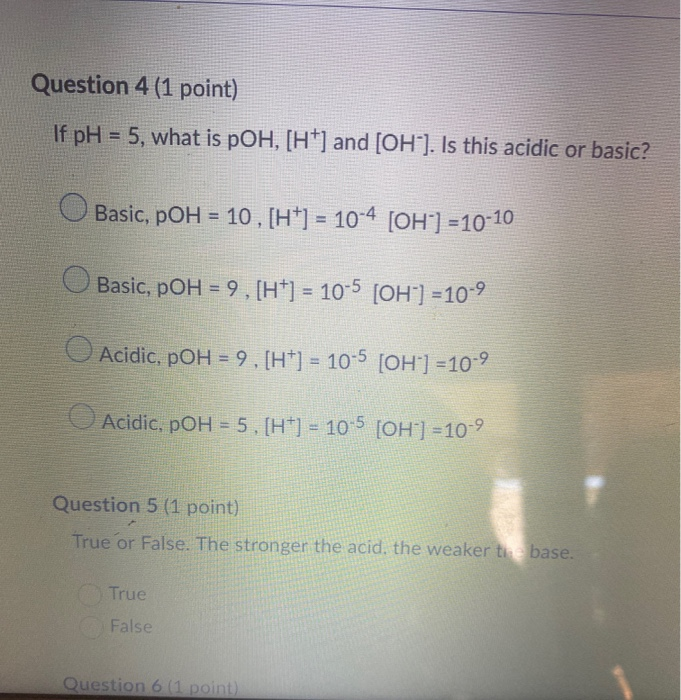
.PNG)