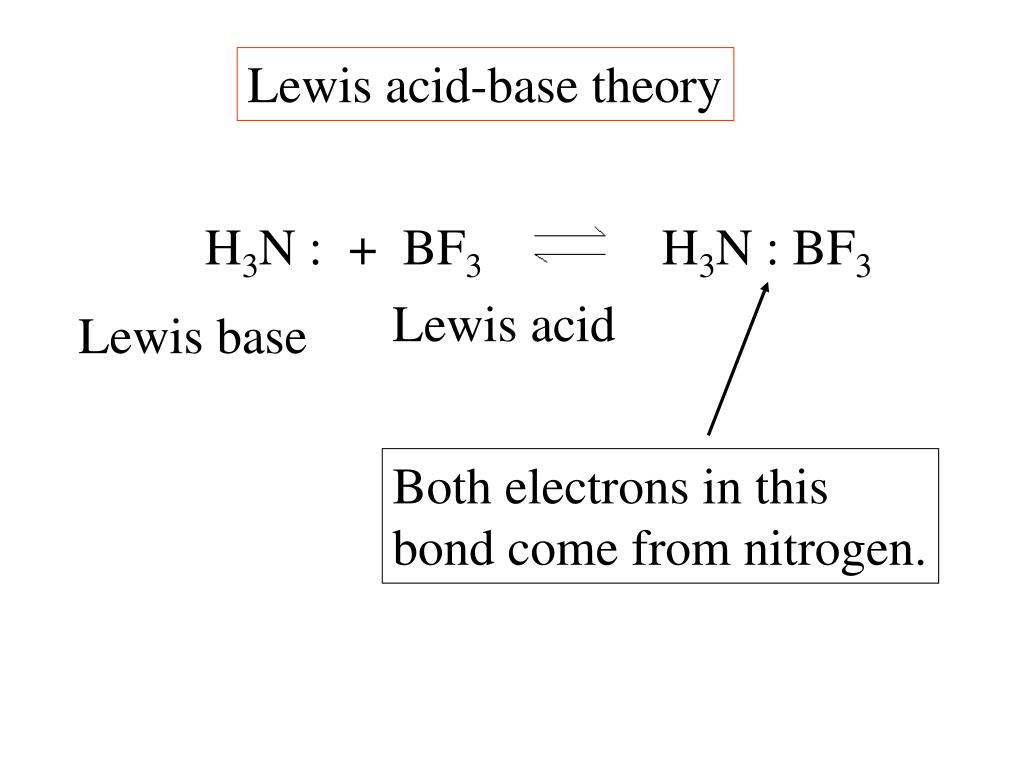
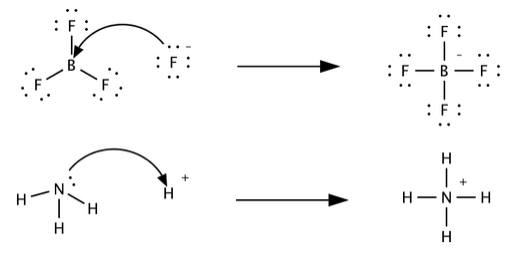
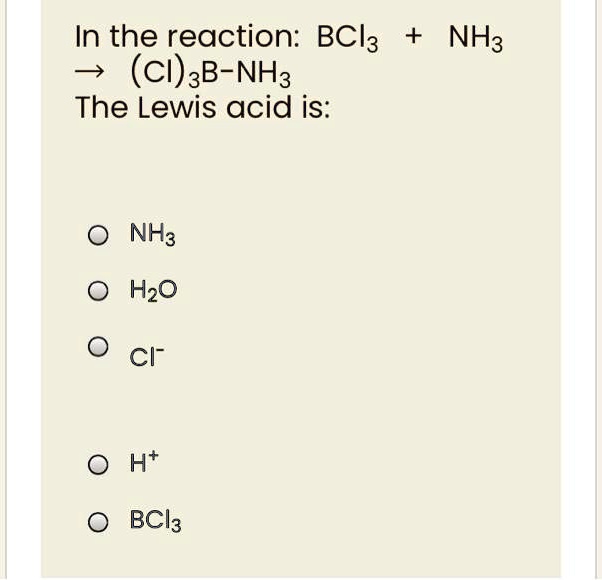
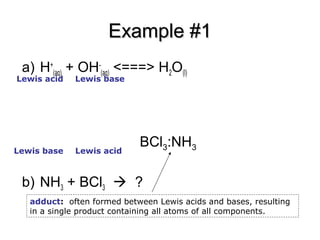
![SOLVED: BCl3 + Cl- = [ BCl 4]- , in the equation which is the base? * a. BCl3 b. Cl c. BCl 4 d. none of these. SOLVED: BCl3 + Cl- = [ BCl 4]- , in the equation which is the base? * a. BCl3 b. Cl c. BCl 4 d. none of these.](https://cdn.numerade.com/ask_previews/39f5fa0a-dd33-4254-bfb2-4ac92f81a9c1_large.jpg)
SOLVED: BCl3 + Cl- = [ BCl 4]- , in the equation which is the base? * a. BCl3 b. Cl c. BCl 4 d. none of these.

BCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle in 2022 | Molecular geometry, Molecular, Vsepr theory
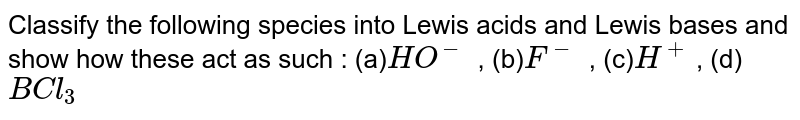
Classify the following species into Lewis acids and bases and show how these act as such: (i) BCl3 (ii) H^(+) (iii) F^(-) (iv) HO^(-)

From σ- to π-Electrophilic Lewis Acids. Application to Selective Organic Transformations | The Journal of Organic Chemistry
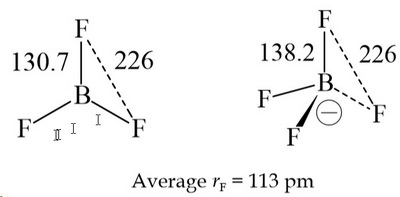
Welcome to Chem Zipper.com......: What are the order of extent back bonding, Lewis acid character and nucleophilicity of (BF3, BCl3, BBr3, BI3)boron trihalides?