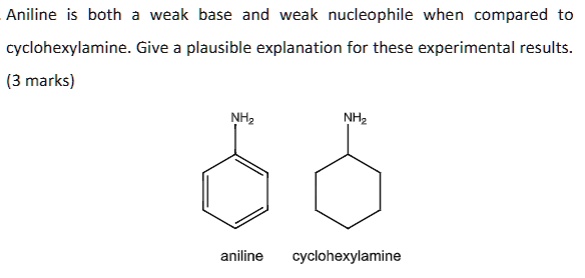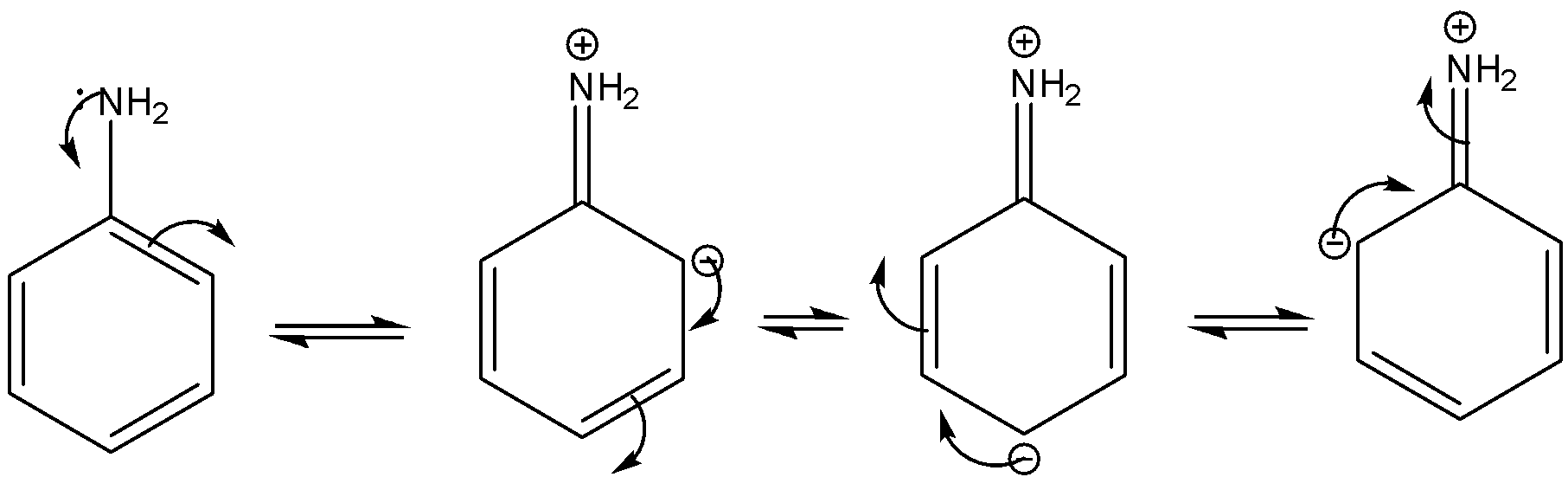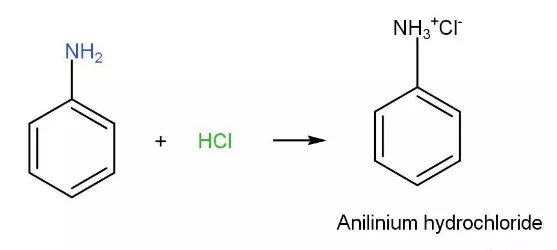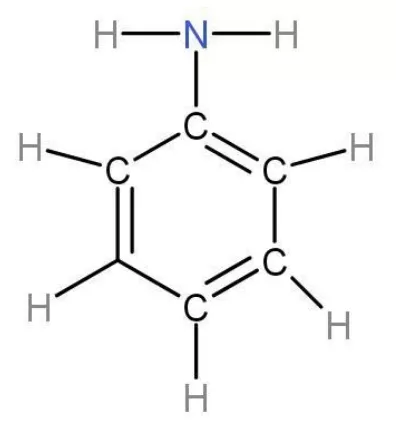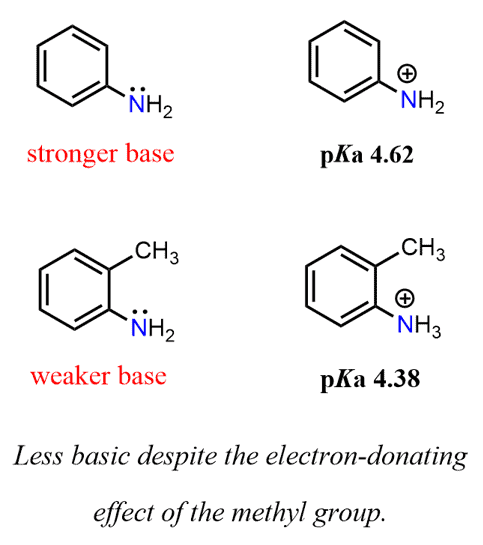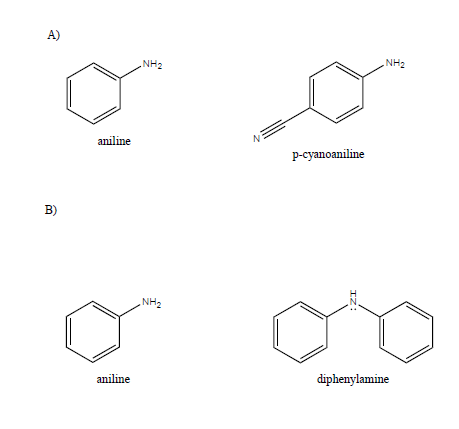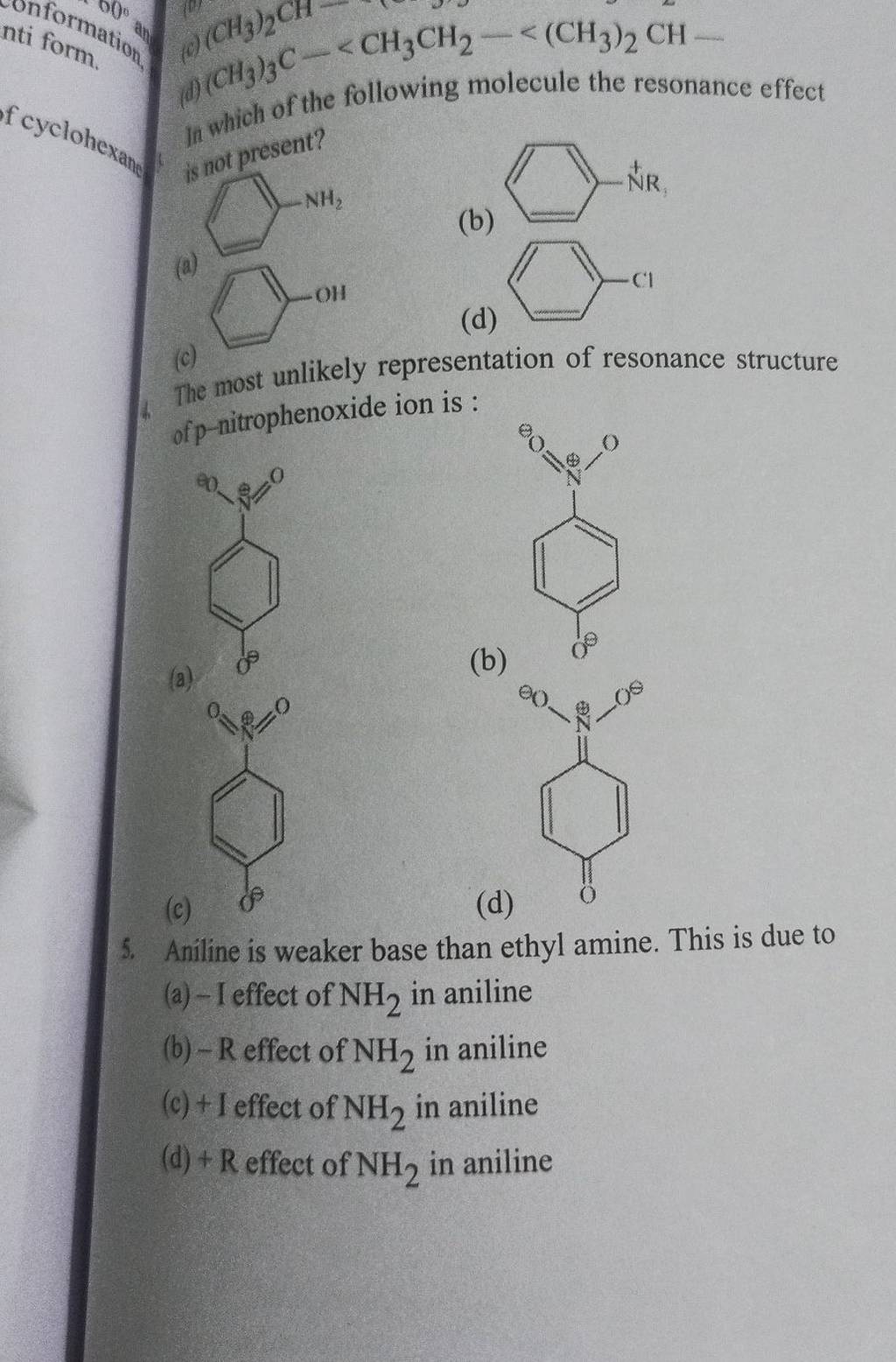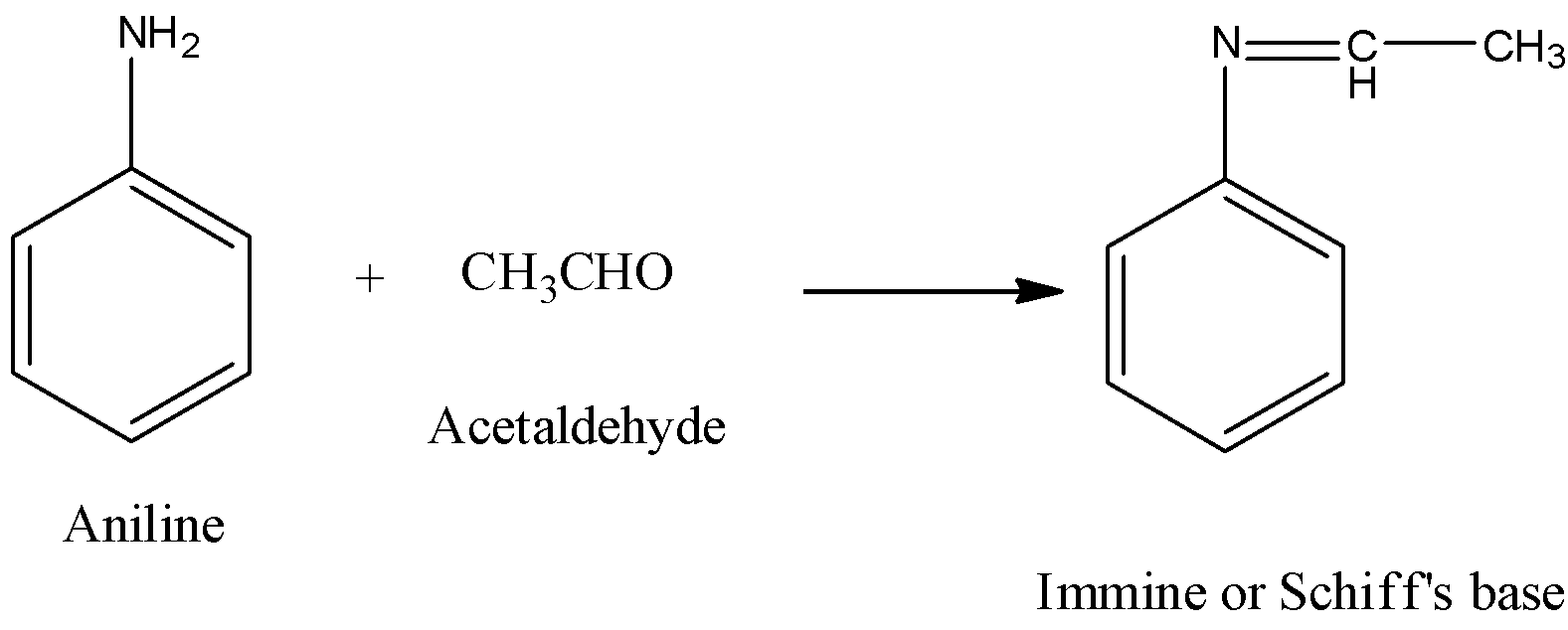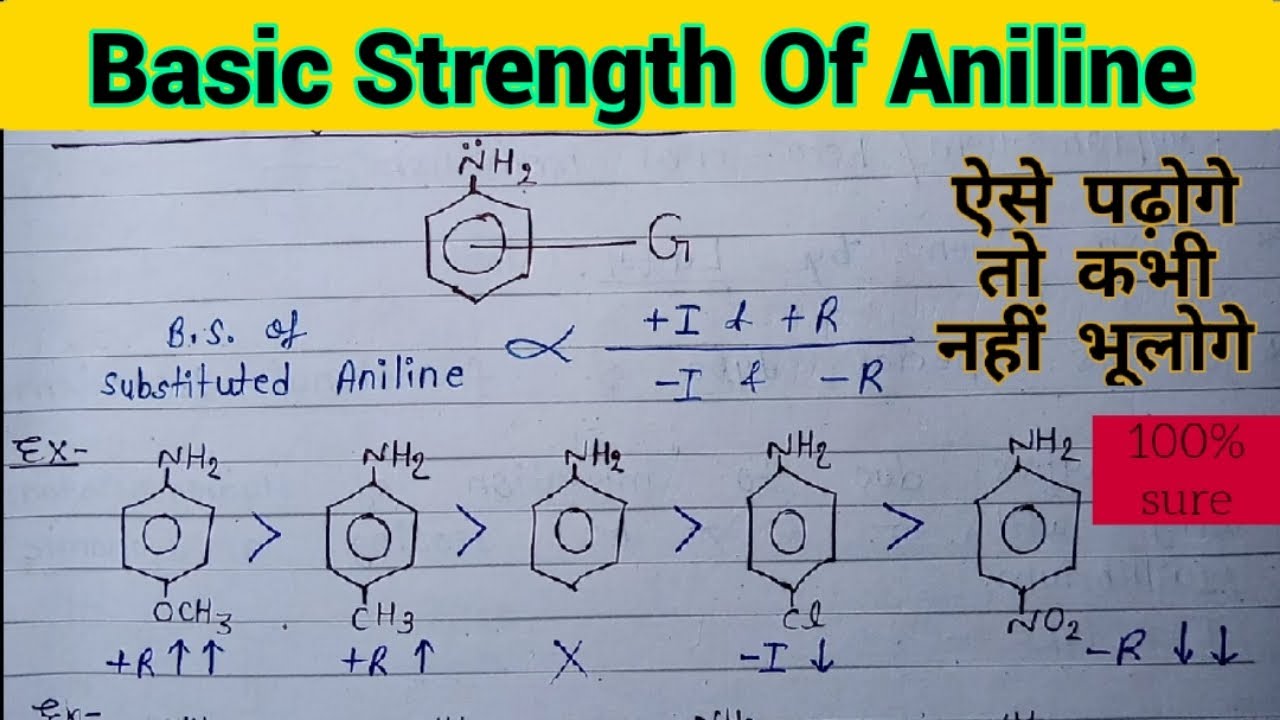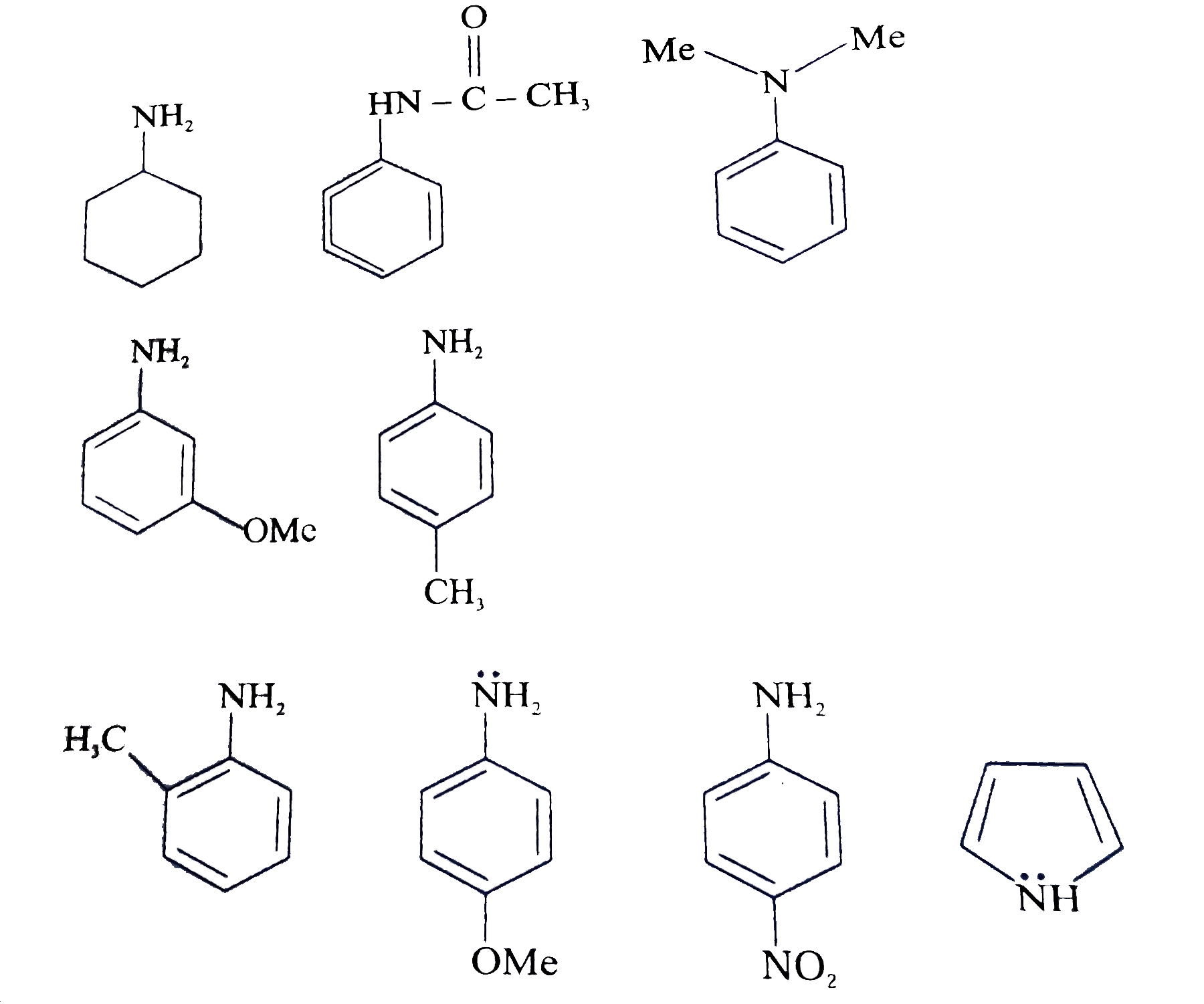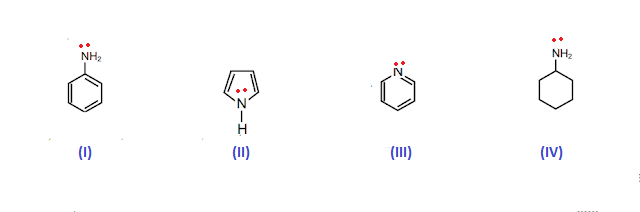
Welcome to Chem Zipper.com......: Arrange in correct order of basic Character of aniline, pyrrol, pyridine and piperidine?
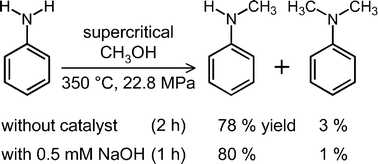
Noncatalytic mono-N-methylation of aniline in supercritical methanol: the kinetics and acid/base effect - Chemical Communications (RSC Publishing)

organic chemistry - Which is the strongest base among the given anilines? - Chemistry Stack Exchange
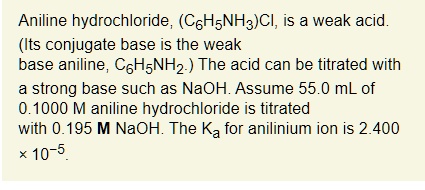
SOLVED: Aniline hydrochloride, (CsHsNH:)CI, is a weak acid. (Its conjugate base is the weak base aniline, CsHsNH2: The acid can be titrated with strong base such as NaOH Assume 55.0 mL of

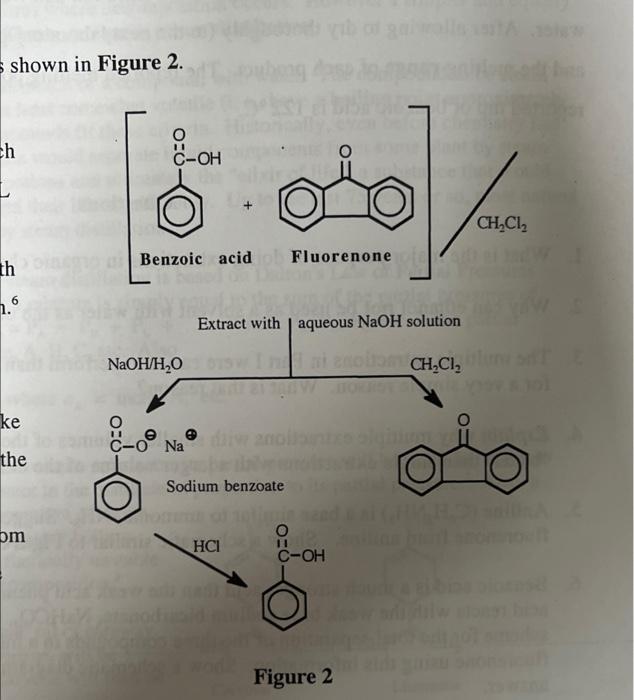
Propose a method to separate the following three compounds: benzoic acid, aniline and naphthalene. How would this separation look in the form of a schematic diagram? | Homework.Study.com

OneClass: Aniline (17), an amine, is soluble in diethyl ether but not water; however, aniline is extr...