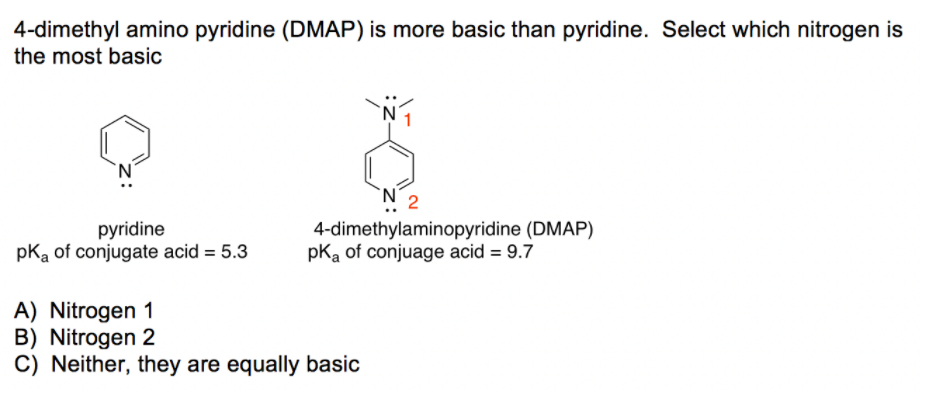
4-(N,N-Dimethylamino)pyridine Hydrochloride as a Recyclable Catalyst for Acylation of Inert Alcohols: Substrate Scope and Reaction Mechanism | Organic Letters

Scheme 3. Reagents and conditions: (a) Ac2O, DMAP, NEt3, THF, RT, 1 h,... | Download Scientific Diagram
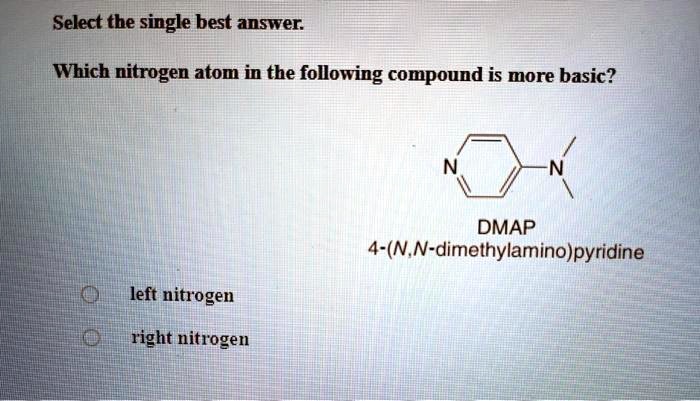
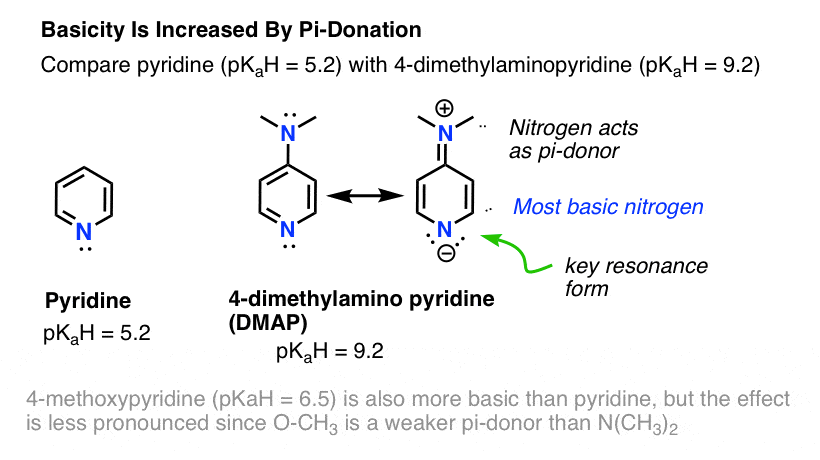
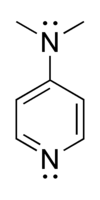
SOLVED: Select the single best answer: Which nitrogen atom in the following compound is more basic? DMAP 4-(N,N-dimethylamino)pyridine left nitrogen right nitrogen

Steric Effects in the Uncatalyzed and DMAP‐Catalyzed Acylation of Alcohols—Quantifying the Window of Opportunity in Kinetic Resolution Experiments - Fischer - 2006 - Chemistry – A European Journal - Wiley Online Library
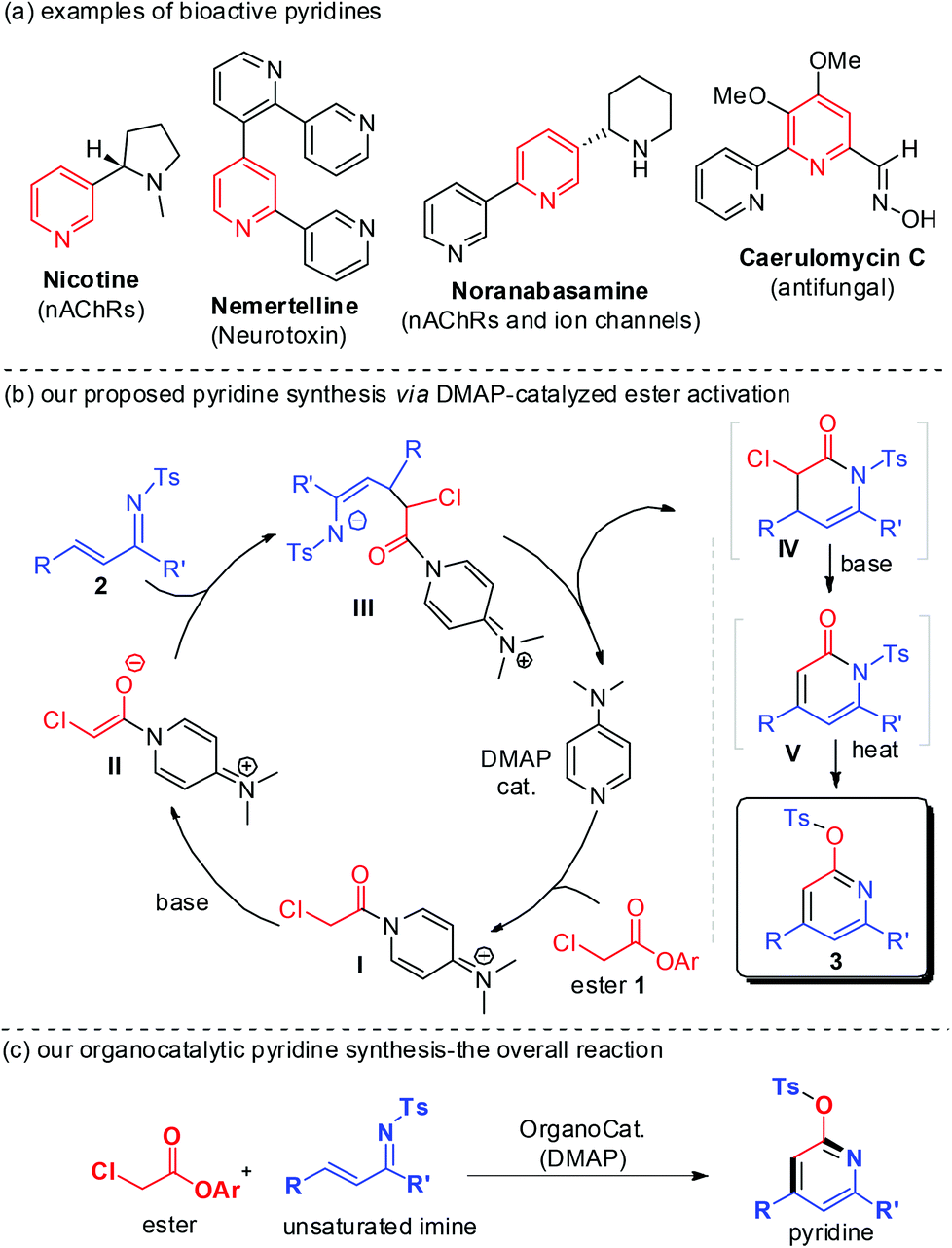
Access to pyridines via DMAP-catalyzed activation of α-chloro acetic ester to react with unsaturated imines - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C3QO00045A

Full article: DMAP-catalyzed cyclization of Schiff bases with α-halo ketones: Synthesis of 1,4-benzoxazines

Fluorous 4‐N,N‐Dimethylaminopyridine (DMAP) Salts as Simple Recyclable Acylation Catalysts - Vuluga - 2010 - Chemistry – A European Journal - Wiley Online Library

Widely Useful DMAP-Catalyzed Esterification under Auxiliary Base- and Solvent-Free Conditions | Journal of the American Chemical Society
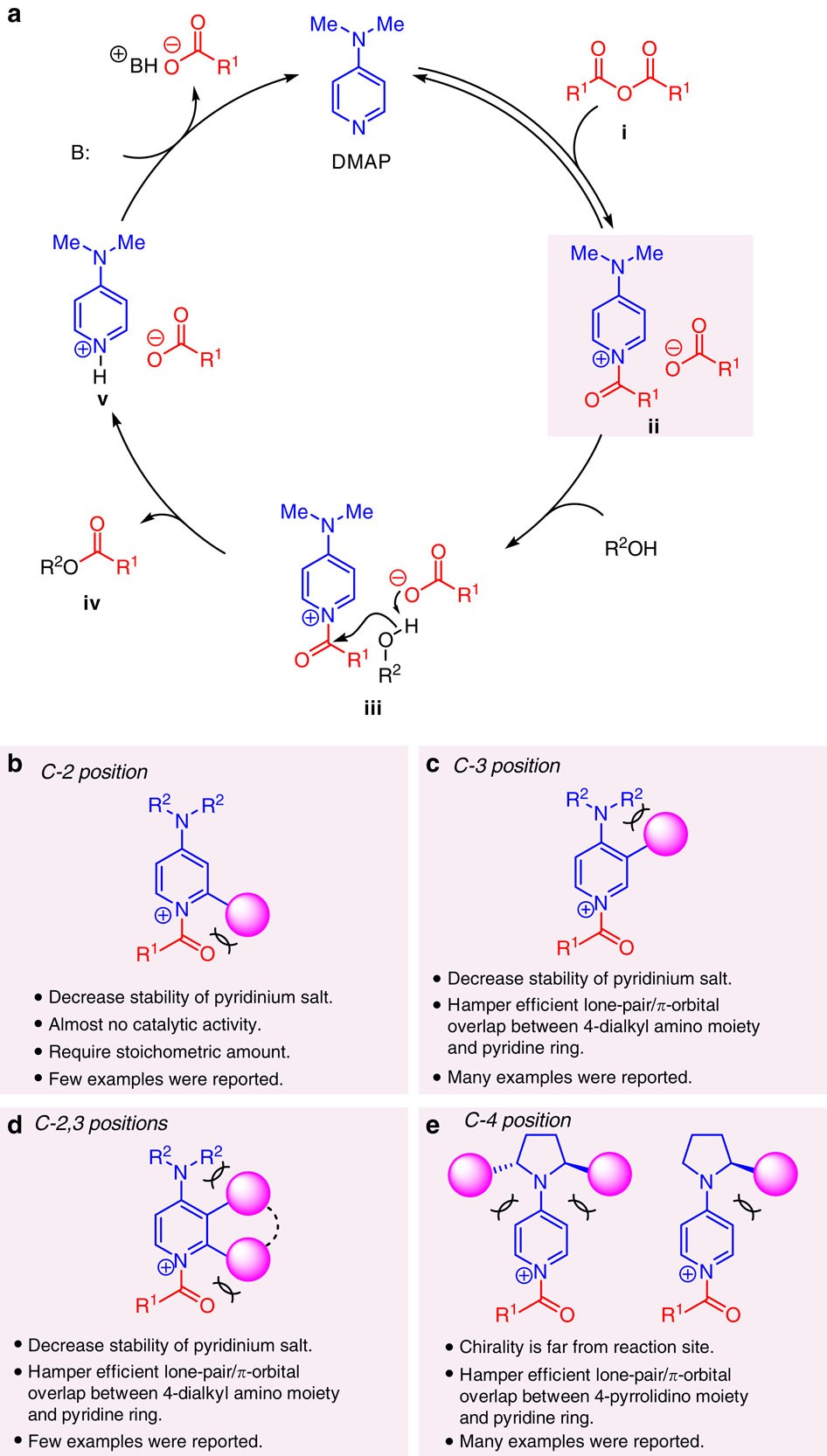
Enantioselective acyl transfer catalysis by a combination of common catalytic motifs and electrostatic interactions | Nature Communications

![PDF] 4-Dimethylaminopyridine as a catalyst in heroin synthesis. | Semantic Scholar PDF] 4-Dimethylaminopyridine as a catalyst in heroin synthesis. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bd2da3cfe7d11eafd93ccc26ca55c536e20d40f0/3-Figure2-1.png)