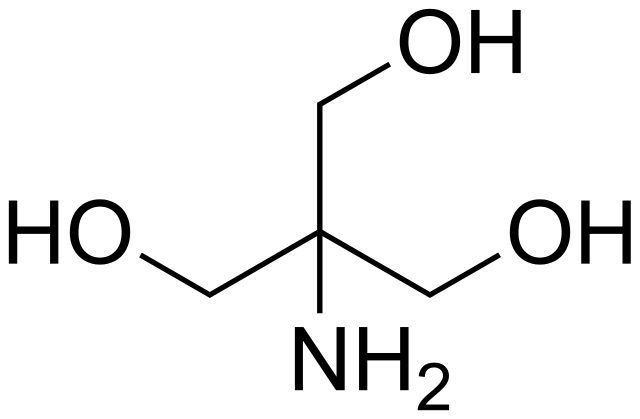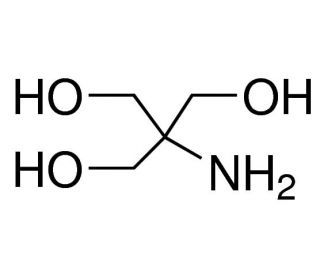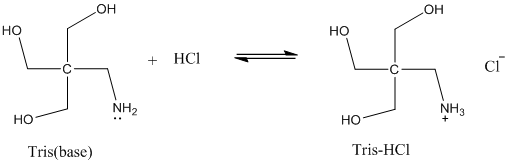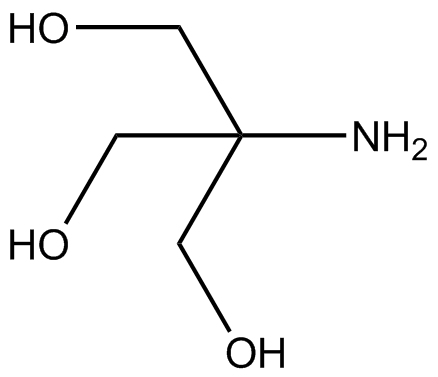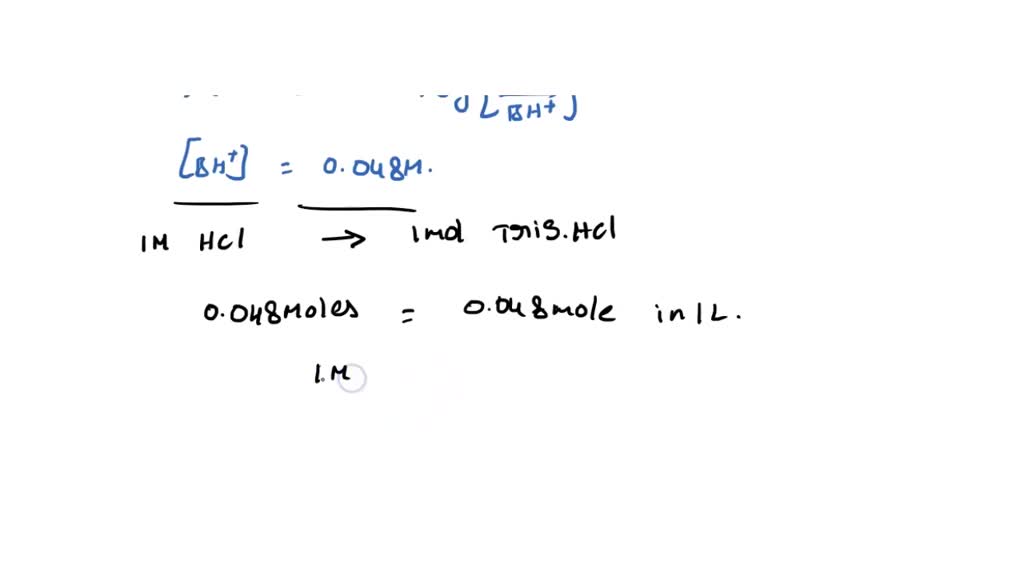
SOLVED: Calculate how you world prepare 100 mL of a 0.1 M TRIS-HCl buffer, pH 7.4 using solid TRIS base (tris-(hydroxymethyl)-aminomethane) (MW 121.1, pKa 8.08). You may find TRIS under several different

New insights into the effect of Tris-HCl and Tris on corrosion of magnesium alloy in presence of bicarbonate, sulfate, hydrogen phosphate and dihydrogen phosphate ions - ScienceDirect

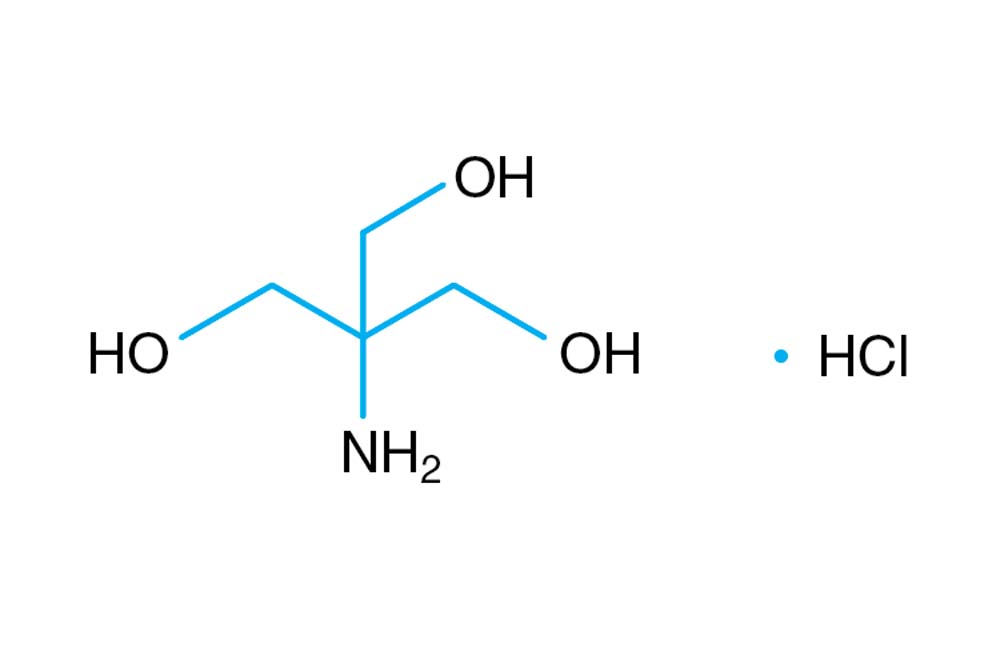
Scheme 2. Structures of Tris, TrisHCl and etilendiaminotetraacetic acid (H4EDTA) : pH and Acid-Base Equilibrium Calculations via a Matrix Representation of Solutions of Acids and/or Bases : Science and Education Publishing

SOLVED: 'Tris' is a buffer often used in biochemistry We can simplify the equilibrium reaction as Tris-HCl+Hz0 Cl- + Hso+ + Tris with pKa = 8.072 and the molar weights m(Tris)-121.135 g/mol
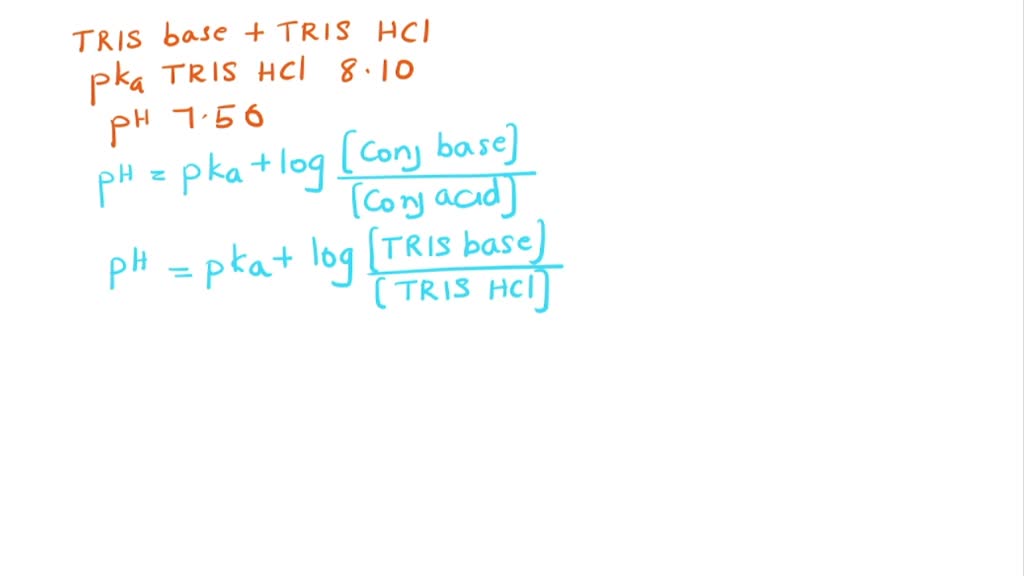

![Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes Tris Base [C4H11NO3] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/tris-base-molecular-weight-calculation-300x204.jpg)